What is rubella?
Rubella is an infection caused by rubella virus. It is also referred to as “German measles” because the rash looks similar to the measles rash and was written about significantly in German medical literature after being first described the 18th century.
What are the symptoms of rubella?
There are two different rubella syndromes: congenital (i.e. newborn) and postnatal (children, adolescents and adults). Postnatal rubella symptoms include low-grade fever, swollen lymph nodes, and rash that starts on the face and spreads to the entire body within 24 hours. The rash lasts 3 days on average. Some patients will have runny nose, cough, headache and/or pink eye prior to onset of the rash. 25-50% of adults with rubella experience no symptoms.
Rubella infection during pregnancy can cause miscarriage, death of the fetus, or Congenital Rubella Syndrome (CRS).
CRS can affect babies’:
- Abdomen (e.g. liver and/or spleen enlargement)
- Development (e.g. developmental disabilities, behavioral disorders, autism)
- Eyes (e.g. cataracts, glaucoma, damage to the retina, vision loss and/or small eyes)
- Growth (e.g. growth restriction)
- Hearing (e.g. sensorineural hearing loss)
- Heart and blood vessels (e.g. patent ductus arteriosus, peripheral pulmonic stenosis)
- Nervous system (e.g. microcephaly AKA small brain, brain inflammation)
- Platelets (e.g. thrombocytopenia or low platelet counts)
- Skin (e.g. “blueberry muffin” lesions)
What are the complications of rubella?
CRS has many complications as noted above. Adolescents and adult women may experience joint pain and inflammation secondary to rubella infection. Brain inflammation (i.e. encephalitis) occurs in 1 in 6000 rubella infections. Low platelet counts (i.e. thrombocytopenia) occur in 1 in 3000 rubella infections.
How is rubella spread?
Rubella can spread from mother to fetus through the placenta during pregnancy, and CRS is most associated with first trimester infections. In older children, adolescents and adults, rubella is spread by inhaling airborne droplets from the cough/sneeze of an infected person. Humans are the only known host of rubella virus. The incubation period (i.e. time from exposure to symptoms) is 2.5 weeks on average. Infected individuals are most contagious a week before to a week after the rash develops. The 25-50% of infected adults who have no symptoms can still spread the virus to others.
Can rubella be treated?
No, unfortunately there is no treatment for rubella. Prevention is, therefore, key.
How common was rubella before introduction of the vaccine?
Prior to introduction of the vaccination in 1969, the last major rubella epidemic was between 1964 and 1965. During this epidemic rubella caused an estimated 12.5 million infections, 11,00 pregnancy losses, 2,100 newborn deaths and 20,000 cases of CRS. In 2023, the last year of available published data, there were 11 reported cases of rubella and 1 reported case of CRS. In 2004, rubella was declared eliminated from the United States, meaning there was no spread within the country due to the high levels of immunity. In 2015 rubella was declared eliminated from all of the Americas. Rare cases still occur in susceptible individuals (e.g. vaccine non-responders, or individuals with weakened immune systems due to medical condition or treatment) from international travel to countries without elimination status.
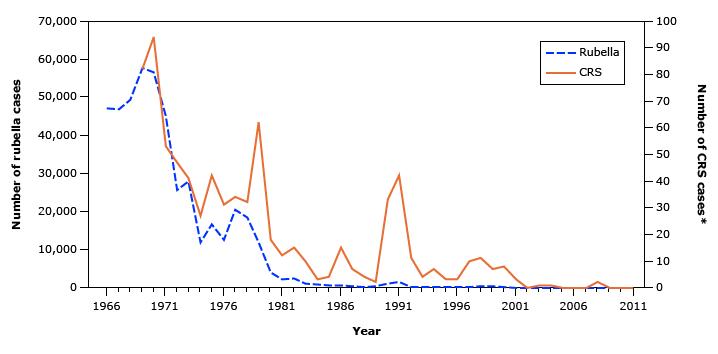
Reproduced from: McLean HQ, Fiebelkorn AP, Temte JL, et al. Prevention of measles, rubella, congenital rubella syndrome, and mumps, 2013: Summary recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 2013; 62:1.
What type of vaccine is the rubella vaccine?
The rubella vaccine is a live, attenuated vaccine. This means that there is actual live virus, but one that has been significantly weakened to the point that it cannot cause actual disease in an individual with a normally functioning immune system. Though weakened so it cannot cause actual disease, this type of vaccine still creates a robust immune system response by our bodies. Attempts to create an inactivated vaccine (i.e. no live virus, like the polio shot) have not produced an adequate protective immune response. The initial live, attenuated vaccine against just rubella was created in the 1969. A video further explaining the process of creating live attenuated vaccines can be found here.
In 1971 the rubella vaccine was combined with individual vaccines for mumps and measles after head-to-head studies showed equal effectiveness and no more side effects with the combined vaccine compared to doing the three components separately. A separate, individual rubella vaccine has not been available in the U.S. in decades.
Note: individuals with a weakened immune system due to medical conditions or because of medical treatment should not receive live, attenuated vaccines unless otherwise specifically recommended by their healthcare professional.
How effective is the rubella vaccine?
A single dose of MMR vaccine is 97% effective for developing lifelong immunity to rubella.
When is the rubella vaccine administered?
The MMR vaccine is administered in two doses. The first dose is typically administered between 12-15 months of age. The second dose is typically given between 4-6 years of age and prior to the start of kindergarten, but can be given any time after 4 weeks have passed from the 12–15-month-old dose.
Maternal antibodies that cross through the placenta and are transferred through breastmilk protect a baby until about 6 months of age. There are special situations in which an infant between 6-12 months of age may receive an additional dose early. This is recommended for infants traveling to areas without rubella elimination status (or locations with active transmission of measles). Infants who receive an MMR vaccine between 6-12 months still need the two-dose series because an infant’s immune system does not typically produce high enough, and long-lasting enough, antibody levels to protect them for the rest of their lives.
While one dose of MMR provides adequate rubella protection (i.e. 97% effective), two doses of MMR are given primarily because it improves measles immunity from 93% to 97%, which is above the desired 95% herd immunity threshold to prevent local spread.
Is the vaccine safe? What are possible side effects of the rubella vaccine?
Yes! The vaccine is safe. Most individuals receiving an MMR vaccine will have no side effects.
The temporary (usually less than 24-48 hours) possible side effects include:
- Localized reactions (such as redness, soreness, swelling and the injection site), up to 40% of recipients
- Drowsiness in 27-45% of recipients
- Decreased appetite in 21-45% of recipients
- High fever 5-12 days after the vaccine is given, up to 35% of recipients
- Mild measles-like rash, less than 5% of recipients. Also typically 1-2 weeks later
- Temporary decrease in platelets (thrombocytopenia), 1-3 cases per 100,000 recipients. Remember, rubella infection itself causes thrombocytopenia in 1 in 3,000 cases
- Febrile seizure in 0.04% of children under age 4 receiving the combined MMR and Chicken Pox vaccine (PAR has not, and does not, administer this combination vaccine). While simple febrile seizures are extremely scary to experience, thankfully they do not cause short- or long-term complications for the brain, slow development, or decrease learning or IQ.
Why do people think the MMR vaccine causes autism, and how do we know that it doesn’t?
In 1998, a British medical journal called The Lancet published an article by Andrew Wakefield claiming that the MMR vaccine increased the risk for autism and colitis (inflamed large intestine). This was a case series in which 12 children were reviewed, and 8 children were diagnosed with autism after the vaccine. Wakefield’s methodologies were later investigated more closely, and the article was eventually retracted in 2010 by The Lancet after investigators found that there were ethical violations (financial gain/conflict of interest) and deliberate fraud with selection/falsification of data. They also found that the children in the study were carefully selected, and Wakefield’s research was funded by lawyers hired by parents involved in lawsuits against vaccine manufacturers.
Since Wakefield’s case series and article, there have approximately 20 large studies in seven different countries in Europe, North America, and Asia. These studies involved hundreds of thousands of children who were grouped into two groups: those who received the MMR vaccine and those who did not. The two groups were matched for multiple variables (healthcare-seeking behavior/accessibility, socioeconomic background, and medical background). All of the studies have shown that there was no difference or increased risk of autism associated with receiving the MMR vaccine.
Why is there a vaccine for rubella?
The benefits are two-fold:
- Every woman vaccinated as a child has protected any and all of her unborn children from the devastating effects of Congenital Rubella Syndrome, rubella-induced miscarriages and fetal deaths.
- Maintain rubella elimination status in the United States. As we have seen with measles in 2025-2026, when vaccination rates drop below herd immunity thresholds, previously eliminated vaccine-preventable illnesses re-emerge.
References
2024. “Rubella”, Red Book: 2024–2027 Report of the Committee on Infectious Diseases, Committee on Infectious Diseases, American Academy of Pediatrics, David W. Kimberlin, MD, FAAP, Ritu Banerjee, MD, PhD, FAAP, Elizabeth D. Barnett, MD, FAAP, Ruth Lynfield, MD, FAAP, Mark H. Sawyer, MD, FAAP
https://www.uptodate.com/contents/rubella
https://wonder.cdc.gov/nndss-annual-summary.html
https://www.chop.edu/vaccine-education-center/vaccine-details/measles-mumps-and-rubella-vaccines
Herrera OR, Thornton TA, Helms RA, Foster SL. MMR Vaccine: When Is the Right Time for the Second Dose? J Pediatr Pharmacol Ther. 2015 Mar-Apr;20(2):144-8. doi: 10.5863/1551-6776-20.2.144. PMID: 25964732; PMCID: PMC4418682.
https://www.cdc.gov/vaccines/hcp/by-disease/mmr.html
Eggertson L. Lancet retracts 12-year-old article linking autism to MMR vaccines. CMAJ. 2010 Mar 9;182(4):E199-200. doi: 10.1503/cmaj.109-3179. Epub 2010 Feb 8. PMID: 20142376; PMCID: PMC2831678.
